 The amino acid composition of the MdPGIP1 protein was calculated using the database. The nucleotide sequence was translated into the encoding polypeptide using the database. The MdPGIP1 gene sequence with the GenBank accession was used during in silico analysis in this study. This further characterization forms part of new information on the MdPGIP1 encoded protein. To further explore the biochemical characteristics of MdPGIP1 as a potential tool in improving disease resistance of food crops, in silico analyses were performed to compare the properties of MdPGIP1 to other characterised PGIPs. In addition, further studies performed to date (Matsaunyane and Oelofse, unpublished) indicate that the protein also inhibits PGs from Verticillium dahlia, Botrytis cinerea, Colletotrichum acutatum and Colletotrichum coccodes, but not that of Fusarium verticillioides. MdPGIP1 inhibits PGs from Botryosphaeria obtuse and Diaporthe ambigua, which are both apple pathogens. The MdPGIP1 gene sequence elicited interest in its potential use as an anti-fungal agent and was subsequently used to transfer into potato and tobacco. The sequence of the MdPGIP1 accession DQ185063 was compared to the MdPGIP accession MDU77041 and the results showed that the two gene sequences share a 100% identity. isolated the complete MdPGIP1 gene from gDNA of M. isolated mRNA containing the complete coding sequence region of the MdPGIP gene from cDNA of M. Knowledge about the structural properties of PGIPs can provide valuable insight into the nature and dynamics of these interactions with fungal PGs, and also helps to identify promising candidate PGIPs for biotechnological approaches to improve plant disease resistance. PGIPs are not specialized inhibitors of a single PG, but rather versatile proteins that capable of recognising different surface motifs of structurally variable PGs. 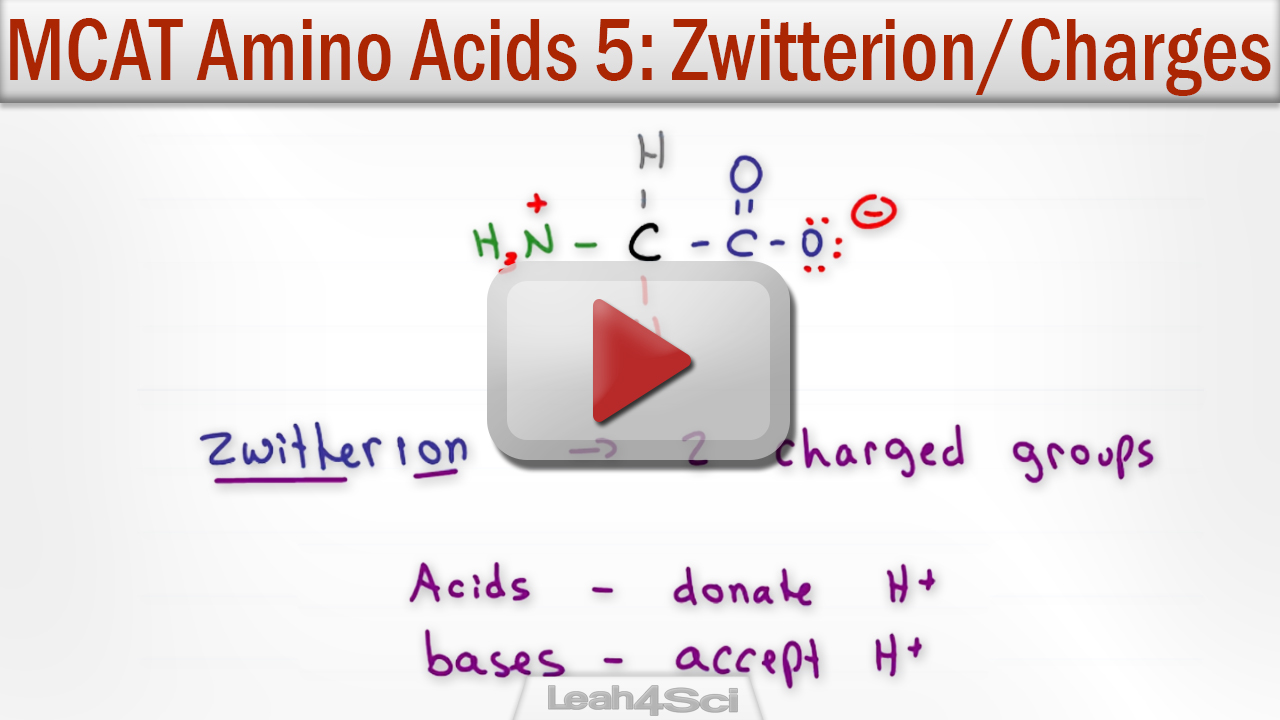
The action of PGIPs on PGs during fungal attack slows down the infection rate and facilitates the prolonged existence of mid-sized oligogalacturonides (damage-associated molecular pattern molecules, DAMPs), which in turn can elicit a general defence response from the plant. PGIPs may specifically inhibit fungal polygalacturonases (PGs). Polygalacturonase inhibiting proteins (PGIPs) are part of the innate immune system of plants. The overall properties of the MdPGIP1 protein is similar to that of the prototypical Phaseolus vulgaris PGIP 2 ( PvPGIP2), and the detected differences supported its use in biotechnological applications as an inhibitor of targeted fungal polygalacturonases (PGs). Furthermore, the secondary and tertiary structure of MdPGIP1 was modelled, and revealed that MdPGIP1 is a curved and elongated molecule that contains sheet B1, sheet B2 and 3 10-helices on its LRR domain. The molecular mass of non-glycosylated MdPGIP1 was calculated as 36.615 kDa and the theoretical isoelectric point as 6.98. The typical consensus glycosylation sequence of Asn-X-Ser/Thr was identified in MdPGIP1, indicating potential N-linked glycosylation of MdPGIP1. The hydropathic evaluation of MdPGIP1 indicated a repetitive hydrophobic motif in the LRR domain and a hydrophilic surface area consistent with a globular protein. In silico characterization of the MdPGIP1 polypeptide revealed domains typical of PGIP proteins, which include a 24 amino acid putative signal peptide, a potential cleavage site for the signal peptide, a 238 amino acid leucine-rich repeat (LRR) domain, a 46 amino acid N-terminal domain and a 22 amino acid C-terminal domain. FindingsĪnalysis of the MdPGIP1 nucleotide sequence revealed that the gene comprises 993 nucleotides that encode a 330 amino acid polypeptide. The gene was used to transform tobacco and potato for enhanced resistance against fungal diseases. 
domestica polygalacturonase inhibiting protein 1 ( MdPGIP1), was isolated from the Granny Smith apple cultivar (GenBank accession no. This gives the following total charge for cysteine at pH 10: Using the Henderson-Hasselbalch in a spreadsheet yields the following $\alpha$ values: Upon deprotonation, the following changes in charge occur for the ionizable groups:Īs an example, let's calculate the charge of cysteine at pH 10. $$\mathrm)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
